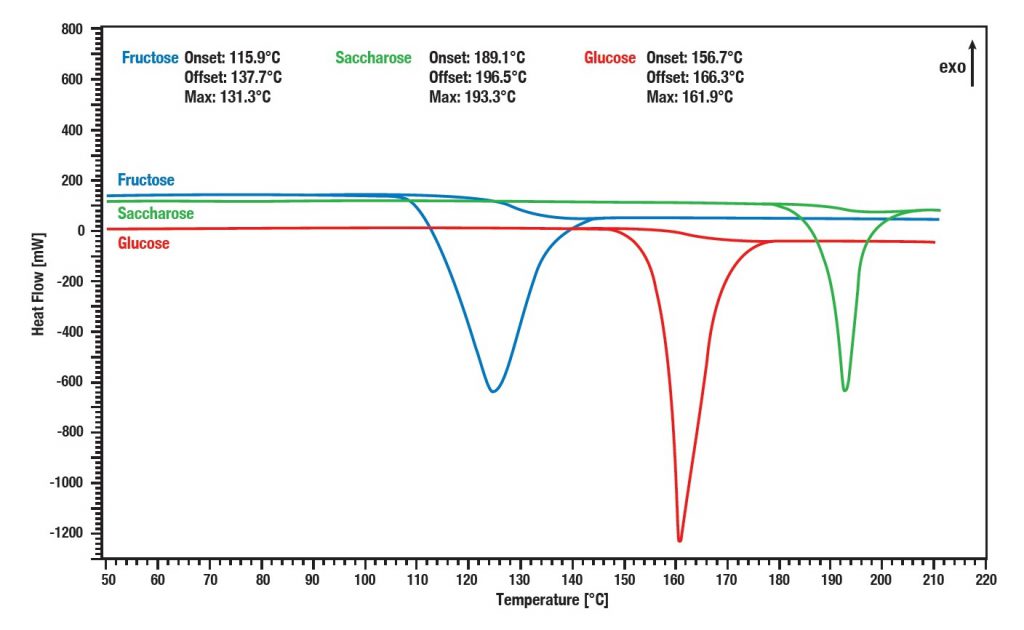
Applications example: Fructose, Glucose and Saccharose
The three evaluated substances (Fructose, Glucose and Saccharose) show distinctive melting points. These melting points can be precisely determined by means of Differential Scanning Calorimetry (DSC). For this the analytical method is frequently used for the determination of unknown substances. Even mixtures with identical molecular weight such as Fructose and Glucose can thus be recognized.
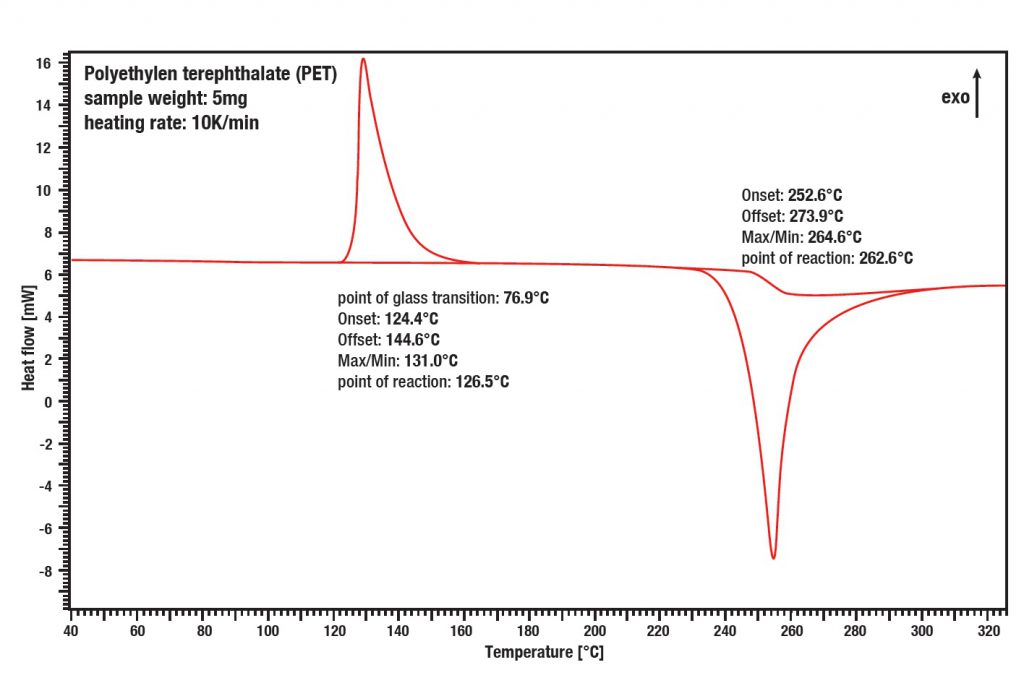
Applications example: Thermoplasts
PolyEthylenTherephtalat (PET) shows a significant endothermic glass point at about 76.9°C, which is quite special for partly crystalline thermoplasts. The relation between the exothermal cold crystallization at 131.0°C and the endothermic melting peak is a measure for the degree of crystallization of the material. In the case of (PET) the crystalline part is very small which results in a good transparency of the material.
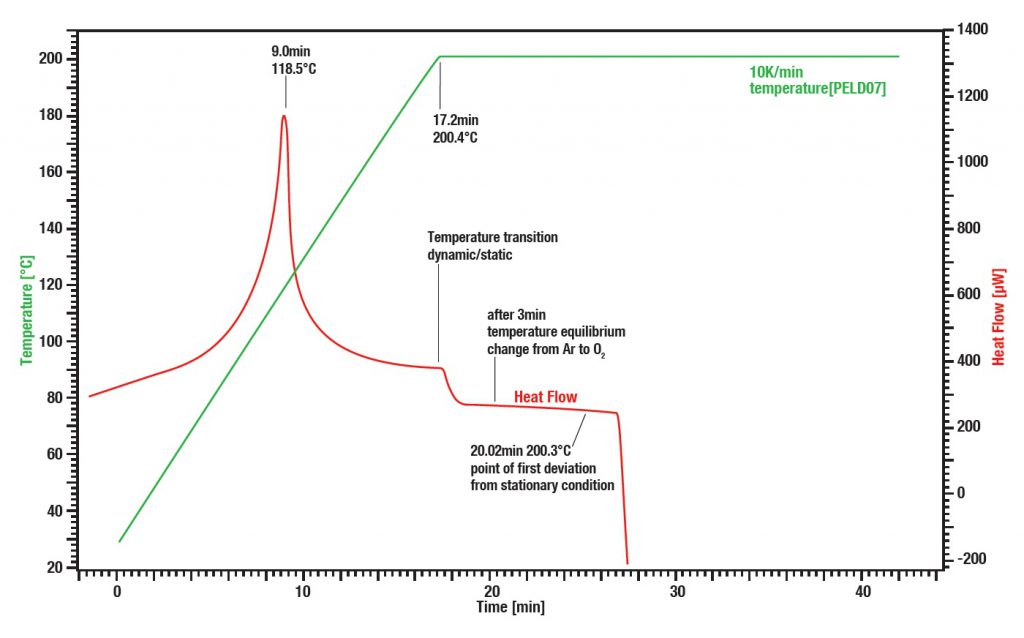
Application example: OIT “Oxidative Induction Time”/ Temperatur
Initially the polyethylene sample is heated up to 200 °C under argon atmosphere with a heating rate of 10 K/min. After 3 minutes at the equilibrium the environment is changed from argon to oxygen. After an additional 5 minutes the exothermal oxidation of the sample starts.
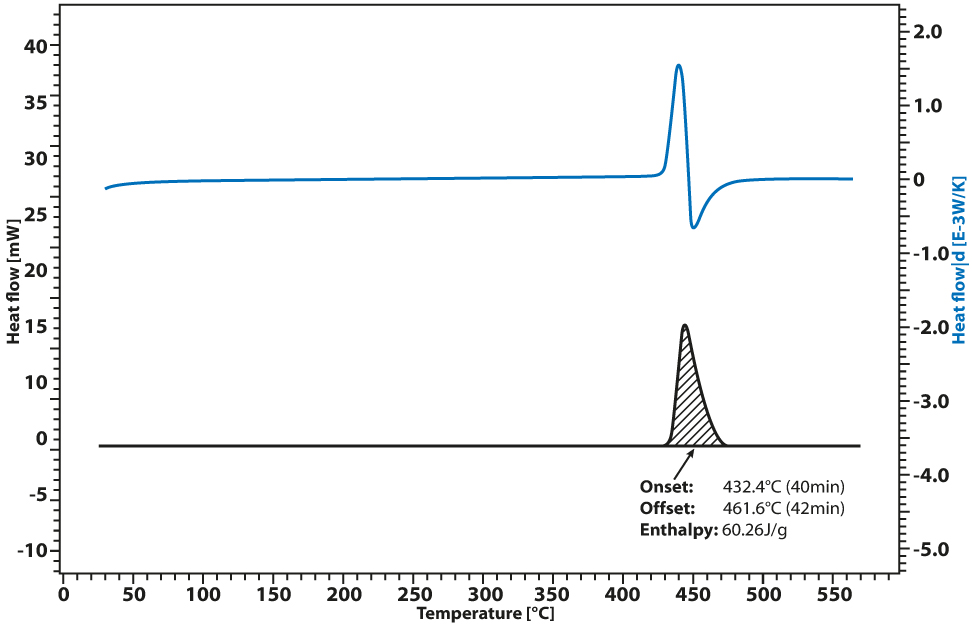
Application example: Auto-ignition of cotton fibers
A sample of cotton fiber, containing inorganic minerals, was measured by DSC PT 1000 to determine the auto ignition temperature (ignition point) and heat of combustion of the cotton content. It is important to distinguish between the flash point and the ignition point.
The flash point describes the temperature at which a substance is inflamed by means of an external ignition source, such as spark, while the auto ignition temperature is the temperature at which a substance inflames without an external ignition source.
The cotton mass sample was heated in the Linseis DSC PT 1000 from room temperature to 600 °C at 10 K/min. The results graph shows the heat flux signal (black curve) and the derivative of the signal (blue curve) as the cotton mass was heated. The cotton mass started to oxidize at 435.4 °C and was completely combusted by 461.6 °C.
The total energy released by the combustion reaction of the cotton mass was 60 J/g as calculated by the area of the heat flux peak using the Linseis evaluation software package.




 Chat Zalo
Chat Zalo





 Đọc thêm
Đọc thêm 



